Thomson was known for his work as a mathematician, where he was recognized as an exceptional talent. The appointment caused considerable surprise, given that candidates such as Osborne Reynolds or Richard Glazebrook were older and more experienced in laboratory work. On 22 December 1884, Thomson was appointed Cavendish Professor of Physics at the University of Cambridge. 
They had two children: George Paget Thomson, who was also awarded a Nobel Prize for his work on the wave properties of the electron, and Joan Paget Thomson (later Charnock), who became an author, writing children's books, non-fiction and biographies. She attended demonstrations and lectures, among them Thomson's. Mary the Less, was interested in physics. Rose Paget, daughter of Sir George Edward Paget, a physician and then Regius Professor of Physic at Cambridge at the church of St. Beginning in 1882, women could attend demonstrations and lectures at the University of Cambridge. In 1890, Thomson married Rose Elisabeth Paget. Thomson received his Master of Arts degree (with Adams Prize) in 1883. He applied for and became a Fellow of Trinity College in 1881. In 1880, he obtained his Bachelor of Arts degree in mathematics ( Second Wrangler in the Tripos and 2nd Smith's Prize). He moved on to Trinity College, Cambridge, in 1876. His parents planned to enroll him as an apprentice engineer to Sharp, Stewart & Co, a locomotive manufacturer, but these plans were cut short when his father died in 1873. Thomson began experimenting with contact electrification and soon published his first scientific paper. In 1870, he was admitted to Owens College in Manchester (now University of Manchester) at the unusually young age of 14 and came under the influence of Balfour Stewart, Professor of Physics, who initiated Thomson into physical research. His early education was in small private schools where he demonstrated outstanding talent and interest in science. Thomson was a reserved yet devout Anglican. He had a brother, Frederick Vernon Thomson, who was two years younger than he was. 
His father, Joseph James Thomson, ran an antiquarian bookshop founded by Thomson's great-grandfather. His mother, Emma Swindells, came from a local textile family. Joseph John Thomson was born on 18 December 1856 in Cheetham Hill, Manchester, Lancashire, England. 
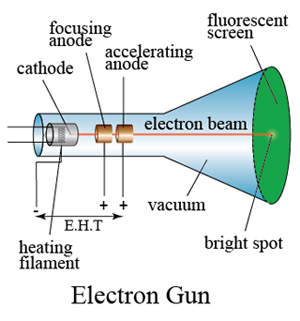
Thomson was also a teacher, and several of his students also went on to win Nobel Prizes. Thomson was awarded the 1906 Nobel Prize in Physics for his work on the conduction of electricity in gases. His experiments to determine the nature of positively charged particles, with Francis William Aston, were the first use of mass spectrometry and led to the development of the mass spectrograph. Thomson is also credited with finding the first evidence for isotopes of a stable (non-radioactive) element in 1913, as part of his exploration into the composition of canal rays (positive ions). In 1897, Thomson showed that cathode rays were composed of previously unknown negatively charged particles (now called electrons), which he calculated must have bodies much smaller than atoms and a very large charge-to-mass ratio. Sir Joseph John Thomson OM FRS (18 December 1856 – 30 August 1940) was a British physicist and Nobel Laureate in Physics, credited with the discovery of the electron, the first subatomic particle to be discovered. NMC cathode Polycrystalline charge distribution heterogeneity local stoichiometry.Owens College (now the University of Manchester) Engineering Ni/Mn/Co distribution in NMC particles may provide a path toward controlling the charge distribution and thus chemomechanical properties of polycrystalline battery particles. The resulting material delivers excellent reversible capacity, rate capability, and cycle life at high operating voltages. The local Mn and Ni concentrations in individual NMC particles are positively and negatively correlated with the electrochemically induced Ni oxidation, respectively, whereas the Co concentration does not impose a clear effect on the Ni oxidation. These NMC particles exhibit a broad, continuous distribution of local Ni/Mn/Co stoichiometry, which does not compromise the global layeredness. Here, we develop heterogeneous compositional distributions in polycrystalline LiNi 1- x- yMn xCo yO 2 (NMC) particles to investigate the interplay between local stoichiometry and charge distribution. However, little is known about how the local TM stoichiometry influences the charging behavior of battery particles thus impacting battery performance. The isostructural nature of Li-layered cathodes allows for accommodating multiple transition metals (TMs).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
